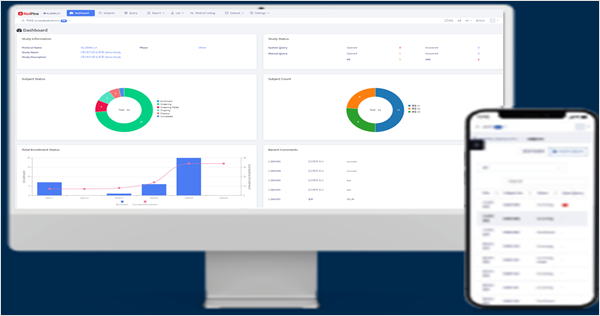
CDMS
Our system ensures research efficiency, data accuracy,
and reliability by adhering to CDISC standards from the design phase of clinical trials.
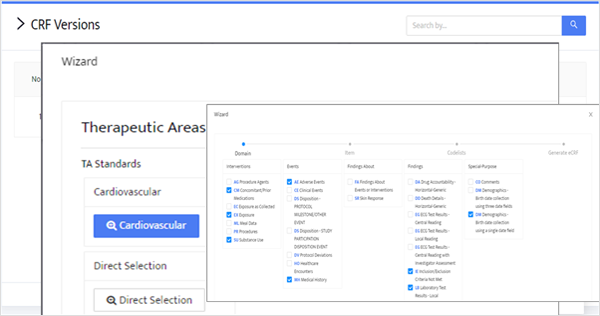
Notably, it facilitates international standard Electronic Data Capture (EDC) setup through
RedPine Builder’s automated functions.
Features

- User-Centered Convenience
-
- User-Centered UI/UX Enhancement
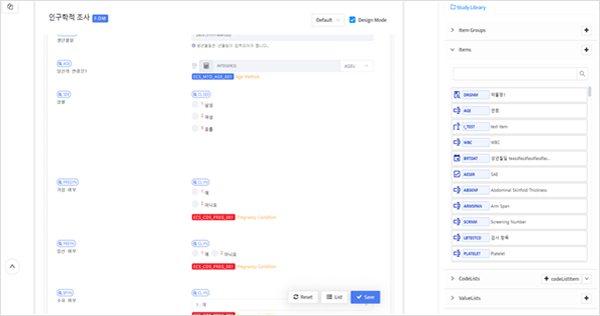
- Drag & Drop Form Design
- Our system supports DEV, UAT, and PROD environments, ensuring version management and robust user testing.
- We achieve improved data quality through various ECS Rules.
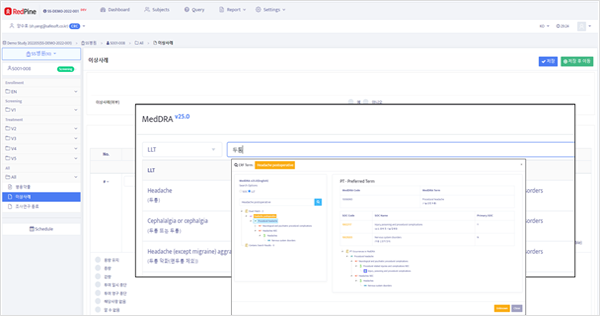
- Users can seamlessly access the Medical Dictionary in both Korean and English.
- Our system provides a download function for Clinical Data Sets, A-CRFs, B-CRFs, and Data CRFs.

- Regulatory Compliance and Quality Control
-
- We adhere to 21 CFR Part 11 (FDA) for clinical trial electronic data processing and management.
- Continuous quality assurance activities are performed by a person in charge with ISO 9001:2015 auditor qualification.
- Our system complies with ISO 27001:2013 Information Security Management Systems.

- Standards and Compatibility
-
- Our system offers an automated setup function through the CDISC SHARE API.
- It supports importing and exporting ODM XML files, including CDISC standard metadata and ECS.
- Our system seamlessly integrates with other EDC systems that also support ODM.
- By adhering to CDISC standards, we ensure research data efficiency, accuracy, and reliability.

- Data Safety and Protection
-
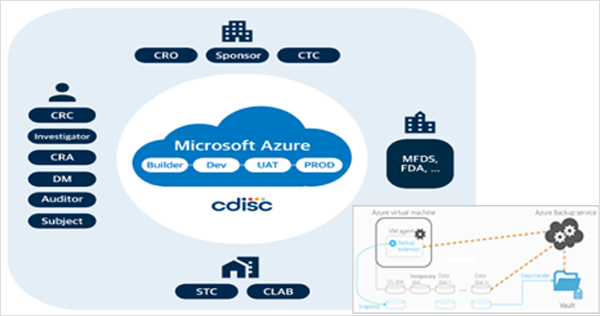
- Microsoft Cloud Azure adheres to major industry standards (ISO/IEC 27001:2013 and NIST SP 800-53).
- Azure Backup ensures data protection through local redundant storage (LRS) and regular backups for recovery.